Paul F. Glenn Laboratories for the Biology of Aging at Stanford University.
Sections for Stanford University
- About Stanford University.
- About Stanford University. Overview.
- Paul F. Glenn Laboratories for the Biology of Aging at Stanford University.
Paul F. Glenn Laboratories for the Biology of Aging at Stanford University
The Glenn Foundation for Medical Research has awarded a $5 million grant to Stanford University to launch a new center on the Biology of Aging. This center is the fourth in the country to be funded by the Glenn Foundation for Medical Research whose goal is “to extend the healthy, productive years of life through research on the biology of aging”.
The core mission of the Paul F. Glenn Laboratories at Stanford is to explore the fundamental mechanisms of the biology of aging, focusing on the mechanisms of mammalian stem cell and tissue aging. Another key focus of the Glenn Laboratories at Stanford University will be to invest in the conceptual and technological advances to conquer new frontiers in aging research.

Director: Thomas A. Rando
> RESEARCH
The average life expectancy currently increases at a rate of more than one year every decade. This dramatic demographic change poses economic challenges, but also provides new opportunities. The opportunities for biological research institutions are to explore the exciting biology of aging, with the future possibility that basic principles revealed by this exploration will result in treatments and interventions that delay the aging process and lead to healthier aging.
Aging
Aging is associated with a striking increase in a wide range of age-dependent diseases, including neurodegenerative diseases, diabetes, and cancer. Even in the absence of diseases, aging is accompanied by the decline of a number of physiological functions, for example a decline in muscle strength and in cognitive function. Fundamental questions arise: is age-dependent decline inevitable? Is there some plasticity to the aging process?
For many decades, aging was not considered to be a regulated process, and just thought to be the by-product of wear-and-tear. But this view has changed, and it is now clear that aging is a plastic process, regulated by a combination of environmental and genetic factors. Importantly, studies in model organisms (yeast, worm, flies, and mice) and in humans have unraveled a series of crucial genes and pathways that are implicated in a conserved manner in regulating longevity. Variations in such genes can extend lifespan by 2-3 fold in lower organisms, and by more than 50% in mammals.
Aging and stem cells
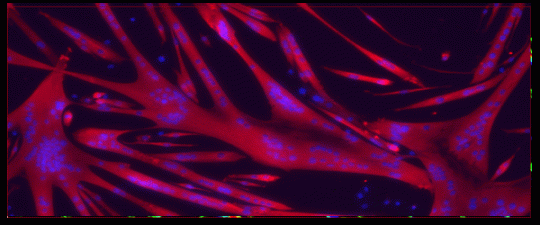
Key aspects of mammalian tissue aging may be attributable to a loss of regenerative capacity of adult stem cells. Unlike differentiated cells, adult tissue-specific stem cells retain a portion of the plasticity of their embryonic counterparts and can differentiate into specific cell types. Adult stem cells have now been identified in most adult tissues in mammals, including the highly regenerative blood, intestine, and skin, as well as the less regenerative skeletal and cardiac muscle and brain. Adult tissue stem cells play important roles in overall tissue homeostasis, repair in response to injury, and in the adaptive nature of the tissue. For example, in the nervous system, adult stem cells are important for adult learning and the formation of new memories.
Throughout the course of organismal lifespan, adult stem cells face the challenge of maintaining an undifferentiated, yet committed state that is primed to respond to the environment. Adult stem cells are subject to the environmental stresses and intracellular damages that accompany the aging process. A fundamental question is whether stem cells progressively lose their potential to self-renew and properly differentiate during organismal aging, and if so, what are the molecular mechanisms underlying this decline. Are age-dependent defects to stem cells reversible?
Future prospects
Understanding the mechanisms by which stem cells and tissue regeneration are affected during aging will give pivotal insights into ways to delay age-dependent decline. Tapping into the regenerative potential of dormant endogenous adult stem cells will likely be a promising avenue to prevent and treat a number of age-dependent diseases characterized by tissue degeneration. The recent ability to generate in vitro pluripotent stem cells from adult patients has opened exciting new paths for exogenous stem cell therapies to treat age-dependent diseases. Understanding how age influences stem cell properties will be critical in implementing new therapies and in promoting healthy aging.
> PROGRAMS
The Glenn Laboratories at Stanford University supports the creation of a program in aging research at Stanford through collaborative grants, fellowships, infrastructure facilities, seminars and symposia.
The goal of The Glenn Laboratories for the Biology of Aging is to promote research in the basic biology of the aging process.
This program will fund proposals for research that examines the fundamental mechanisms of aging and longevity.
The primary goal of these grants is to create opportunities for faculty to develop pilot projects that are currently unfunded and that have the potential to provide preliminary data for applications for extramural funding.
> PEOPLE
>> DIRECTOR
Thomas A. Rando MD, PhD
 The Rando laboratory studies the basic molecular mechanisms of stem cell biology, including the regulation of quiescence, activation, proliferative expansion, and progenitor cell fate. A major focus of the research is on age-related declines of stem-cell mediated tissue homeostasis, repair, and regeneration and on the role of the systemic environment of aged organisms in suppressing stem cell functionality. In this context, the laboratory focuses on the biology of muscle stem cells and comparisons with adult stem cells in other tissues. The Rando laboratory also studies both pathogenesis and experimental therapeutics for the muscular dystrophies, with a particular emphasis on stem cell therapies.
The Rando laboratory studies the basic molecular mechanisms of stem cell biology, including the regulation of quiescence, activation, proliferative expansion, and progenitor cell fate. A major focus of the research is on age-related declines of stem-cell mediated tissue homeostasis, repair, and regeneration and on the role of the systemic environment of aged organisms in suppressing stem cell functionality. In this context, the laboratory focuses on the biology of muscle stem cells and comparisons with adult stem cells in other tissues. The Rando laboratory also studies both pathogenesis and experimental therapeutics for the muscular dystrophies, with a particular emphasis on stem cell therapies.
http://med.stanford.edu/profiles/Thomas_Rando/
http://www.stanford.edu/~casco/
>> ASSOCIATE DIRECTORS
Steven E. Artandi MD, PhD
 The Artandi laboratory is interested in how telomere shortening influences cancer, stem cell function, and aging. Telomeres are nucleoprotein complexes that protect chromosome ends and shorten with cell division and aging. The Artandi laboratory has found that telomerase, the reverse transcriptase that synthesizes telomere repeats, also regulates stem cells. The Artandi laboratory is pursuing the function of telomerase through diverse genetic and biochemical approaches.
The Artandi laboratory is interested in how telomere shortening influences cancer, stem cell function, and aging. Telomeres are nucleoprotein complexes that protect chromosome ends and shorten with cell division and aging. The Artandi laboratory has found that telomerase, the reverse transcriptase that synthesizes telomere repeats, also regulates stem cells. The Artandi laboratory is pursuing the function of telomerase through diverse genetic and biochemical approaches.
http://med.stanford.edu/profiles/Steven_Artandi/
http://www.stanford.edu/group/artandi/
…
Anne Brunet, PhD
 The Brunet laboratory studies the molecular basis of longevity. The Brunet laboratory is interested in the mechanism of action of known longevity genes, including FOXO transcription factors and SIRT deacetylases. The Brunet lab focuses on the role of these longevity genes in the mammalian nervous system, particularly in aging neural stem cells. The Brunet laboratory is also discovering novel genes and processes involved in aging using two model systems, the invertebrate C. elegans and an extremely short-lived vertebrate, the African killifish N. furzeri.
The Brunet laboratory studies the molecular basis of longevity. The Brunet laboratory is interested in the mechanism of action of known longevity genes, including FOXO transcription factors and SIRT deacetylases. The Brunet lab focuses on the role of these longevity genes in the mammalian nervous system, particularly in aging neural stem cells. The Brunet laboratory is also discovering novel genes and processes involved in aging using two model systems, the invertebrate C. elegans and an extremely short-lived vertebrate, the African killifish N. furzeri.
http://med.stanford.edu/profiles/Anne_Brunet/
http://www.stanford.edu/group/brunet/
> CONTACT
Thomas A. Rando, MD, PhD
Professor of Neurology and Neurological Sciences
Director of the Glenn Laboratories for the Biology of Aging at Stanford University
Building 100, Room B4-123
VAPAHCS
3801 Miranda Ave
Palo Alto, California 94304
Tel: (650) 849-0444
Fax: (650) 858-3935
Email: rando@stanford.edu
Sheri Jungers
Program Coordinator
Building 100, Room B4-123
VAPAHCS
3801 Miranda Ave
Palo Alto, California 94304
Tel: (650) 849-1999
Fax: (650) 849-1998
Email: sjungers@stanford.edu
###
* Stanford University Medical Center integrates research, medical education and patient care at its three institutions – Stanford University School of Medicine, Stanford Hospital & Clinics and Lucile Packard Children’s Hospital.
** The above information is adapted from materials provided by Stanford University School of Medicine
________________________________________________________________