Method Quickly Assesses Antibiotics
Method Quickly Assesses Antibiotics
Researchers developed a method to quickly identify antibiotics that can treat multidrug-resistant bacteria. The approach can also reveal how these bacteria-killing medications work.
Decades of widespread antibiotic use have encouraged the spread of bacteria with resistance to multiple antibiotics. Many diseases—including tuberculosis, gonorrhea, and Staphlycoccus aureus infections (“staph”)—can be nearly impossible to treat when they’re caused by multidrug-resistant bacteria. Some of these bacteria are resistant to just about every known class of antibiotic.
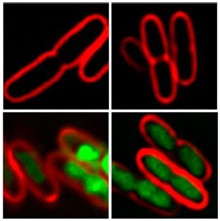
By comparing the appearance of bacterial cells treated with different antibiotic compounds, researchers can determine their likely mode of action./ Image by the researchers, courtesy ofPNAS.
Antibiotics disrupt bacteria’s ability to grow and replicate. Different classes of antibiotics do this through different mechanisms. Some inhibit the creation or repair of the cell wall. Others interfere with the cell’s ability to make protein or replicate DNA. To combat multidrug-resistant bacteria, researchers are searching for new classes of antibiotics that work by different mechanisms than current drugs.
A team led by Drs. Joseph Pogliano and Kit Pogliano of the University of California, San Diego, tested whether they could quickly identify an antibiotic’s class, or mechanism of action, by analyzing the target bacterial cell’s shape and appearance. Earlier NIH-funded research suggested that the structure and appearance of bacteria might provide clues to how these compounds kill cells.
The researchers first treated cultures of Escherichia coli bacteria with antibiotics that have already been classified and well studied. They took various measurements of the cells’ shapes and appearance under fluorescence microscopy with different stains. The study was funded by NIH’s National Institute of General Medical Sciences (NIGMS) and National Institute of Allergy and Infectious Diseases (NIAID). It appeared online on September 17, 2013, in Proceedings of the National Academy of Sciences.
Antibiotics known to be in the same class could be grouped together using the method, termed bacterial cytological profiling (BCP). For instance, compounds that inhibit the bacteria’s ability to make proteins grouped separately from those that inhibited the cells’ ability to replicate DNA. Furthermore, within these class-specific groups, BCP could distinguish smaller subgroups based on the specific molecular mechanisms. For example, cell-wall synthesis inhibitors fell into 3 different groups depending on which stage of the process they inhibited.
To test whether BCP could distinguish the pathways targeted by different antibiotics, the scientists tested 18 known compounds. They put the compounds into 3 separate groups of 10 and blindly exposed E. coli cells to each one. BCP correctly assigned all 30 compounds to their correct cellular targets.
Finally, the team used BCP to determine the previously unknown mechanism of spirohexenolide A, an antibiotic known to be effective against methicillin-resistant S. aureus (MRSA). Follow-up experiments supported BCP’s prediction that spirohexenolide A works by disrupting the membranes of target bacteria.
“Our new method represents the first time that a single test can be performed and identify the likely mechanism of action for a new compound,” Joseph Pogliano says. “This method will allow us to more quickly identify chemicals that kill bacteria, which will accelerate the development of new medicines.”
By Katherine Wendelsdorf, Ph.D.
###
* The above story is reprinted from materials provided by National Institutes of Health (NIH)
** The National Institutes of Health (NIH) , a part of the U.S. Department of Health and Human Services, is the nation’s medical research agency—making important discoveries that improve health and save lives. The National Institutes of Health is made up of 27 different components called Institutes and Centers. Each has its own specific research agenda. All but three of these components receive their funding directly from Congress, and administrate their own budgets.