Discovery opens new window on development, and maybe potential, of human egg cells
Discovery opens new window on development, and maybe potential, of human egg cells
Egg cells hold the key to many mysteries about reproduction, and knowing about the genetic makeup of individual eggs is important in fertility treatment. A new method of looking at genetic material egg cells discard offers a way to learn more about individual eggs without destroying them.
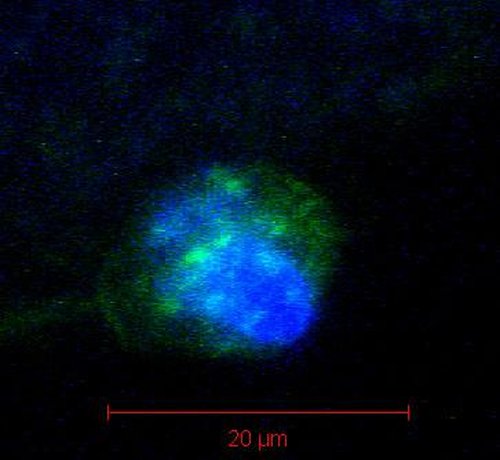
Answers are in the stars To perfect a technique for analyzing mRNA in the polar bodies of egg cells, researchers studied starfish, which produce millions of easily available eggs. The image shows a starfish polar body with stained membrane (green) and DNA (blue). Credit: Carson Lab / Brown University
PROVIDENCE, R.I. [Brown University] — Fertility procedures such as in vitro fertilization (IVF) require a couple and the doctor to place the risky bet that the multiple eggs they choose to fertilize will produce an embryo that will thrive in the uterus. Researchers cannot biopsy eggs directly because that would destroy them, but a new discovery by professors at Brown University and Women & Infants Hospital could lead to new insights about how eggs develop and ultimately inform judgments about how the embryos they produce will fare. The idea is to examine the genetic material the egg cells discarded when they were first forming, to see which genes they were expressing.
“This opens up a whole new time of life for investigation,” said Sandra Carson, professor of obstetrics and gynecology at the Warren Alpert Medical School of Brown University and director of reproductive endocrinology and infertility at Women & Infants.
Oocytes, or eggs, carry half as much genetic material as other cells in the body because a sperm is supposed to donate the other half of the needed DNA. When an oocyte is formed, it casts off a copy of its DNA into a cellular byproduct called a “polar body.” For years, fertility doctors have looked at the DNA in polar bodies for insight into whether the egg would thrive, but until now, nobody had ever found any copies of the oocyte’s messenger RNA (mRNA), the translated messages of genetic code that are tell-tale signs of which genes are active in a cell. Moreover, no one understood how they could detect mRNA if it was there.
“This research gives us a new technique that might prove useful for looking at how genes are being interpreted by the oocyte,” said Peter Klatsky, a research fellow in Carson’s lab, who will present the research Oct. 25 at the American Society for Reproductive Medicine annual meeting in Denver. “This may in the future allow us to ask questions about whether an egg is healthy and therefore whether or not that egg, once fertilized will develop into a healthy baby.”
Along with Gary Wessel, professor of molecular and cellular biology biochemistry at Brown, Klatsky and Carson reasoned that if each polar body did carry mRNA like the oocyte that spawned it, that would be the next best thing to looking for mRNA in the oocyte itself, which is too destrucive. Polar bodies, they hypothesized, could be a reliable and expendable indicator of gene expression in the egg, at least at one key stage in its development.
“Our hypothesis was that along with the discarded DNA, there is cytoplasm and in that cytoplasm there could be information in the form of mRNA and that information could tell us what’s going on in that oocyte,” Klatsky said.
In a series of experiments with donated human oocytes and polar bodies, the trio succeeded in becoming the first to detect tiny amounts of mRNA in polar bodies. Furthermore, they were able to show that the abundance of mRNA in each egg cell correlated with their ability to find it in the polar body, suggesting that what’s expressed in the egg is present in the polar body.
“Now that we’ve figured out that you can detect it, the next question is does it tell you something about the health of the egg,” Klatsky said.
Supporting cast of stars
Achieving these results was no easy task. The amount of mRNA is so small, on the order of quadrillionths of grams, that the team had to develop a new procedure for amplifying it using polymerase chain reaction, a method of making copies of DNA. A key step was to break with tradition and not try to isolate mRNA to amplify it. Instead, Wessel said, they took steps to retool the polymerase chain reaction process to find the mRNA itself.
To perfect the technique, the team practiced on sea stars (also known as starfish) that Wessel has long studied in his basic biological research on fertilization. At the single-cell level of eggs, sea stars work much like people, Wessel said, but they produce a lot more eggs and polar bodies and those are much easier to study.
“Starfish have been amazingly important for understanding how oocytes develop to become fertilizable,” Wessel said. “We can get a few or a dozen eggs from people each month but a starfish has about 10 million eggs.”
With an interest in fertility, Wessel has long kept in touch with clinicians working with humans at Women & Infants. Carson directs those efforts — Klatsky is a fellow in her division — and so they all forged a collaboration.
Administrators backed them up. One measure of how risky their hypothesis was is that all $100,000 of funding for their research came from internal sources: seed grants awarded from the Office of the Provost at Brown University and from the Center of Excellence in Women’s Health at Women & Infants Hospital.
Now that the gamble has paid off in mRNA, the team is pushing ahead to find out whether it can inform both the basic understanding of eggs, and the ultimate promise of improving fertility treatment.
* The above story is reprinted from materials provided by Brown University
** More information at Brown University (Providence, Rhode Island, USA)